LEO the lion says GER!
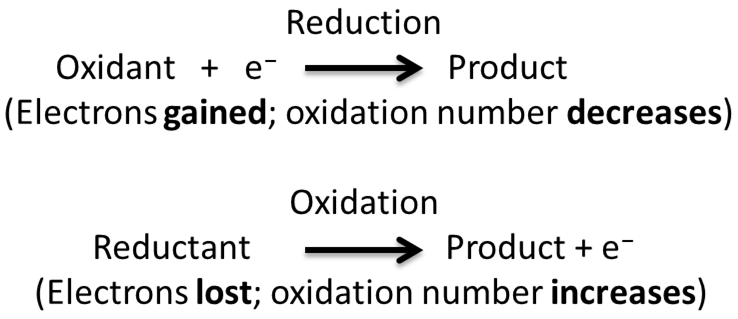
Loses Electrons Oxidation reaction
Gains Electrons Reduction reaction
CuS + HNO3 -> Cu SO4 + NO (g) + H2O (equation not balanced).
3CuS + 8HNO3 -> 3 CuSO4 + 8NO(g) + 4H2O (equation balanced)
3CuS2+ + 3S2- + 8H+ + 8NO3- -> 3Cu2+ + 3SO42- + 8NO(g) + 4H2O (equation written in ionic nomenclature)
Loses Electrons Oxidation reaction
Gains Electrons Reduction reaction
CuS + HNO3 -> Cu SO4 + NO (g) + H2O (equation not balanced).
3CuS + 8HNO3 -> 3 CuSO4 + 8NO(g) + 4H2O (equation balanced)
3CuS2+ + 3S2- + 8H+ + 8NO3- -> 3Cu2+ + 3SO42- + 8NO(g) + 4H2O (equation written in ionic nomenclature)